Wu 2022 Neuromolecular Med
| Wu Z, Ho WS, Lu R (2022) Targeting mitochondrial oxidative phosphorylation in glioblastoma therapy. Neuromolecular Med 24:18-22. https://doi.org/10.1007/s12017-021-08678-8 |
Wu Z, Ho WS, Lu R (2022) Neuromolecular Med
Abstract: As a multi-functional cellular organelle, mitochondrial metabolic reprogramming is well recognized as a hallmark of cancer. The center of mitochondrial metabolism is oxidative phosphorylation (OXPHOS), in which cells use enzymes to oxidize nutrients, thereby converting the chemical energy to the biological energy currency ATPs. OXPHOS also creates the mitochondrial membrane potential and serve as the driving force of other mitochondrial metabolic pathways and experiences significant reshape in the different stages of tumor progression. In this minireview, we reviewed the major mitochondrial pathways that are connected to OXPHOS and are affected in cancer cells. In addition, we summarized the function of novel bio-active molecules targeting mitochondrial metabolic processes such as OXPHOS, mitochondrial membrane potential and mitochondrial dynamics. These molecules exhibit intriguing preclinical and clinical results and have been proven to be promising antitumor candidates in recent studies.
• Bioblast editor: Gnaiger E
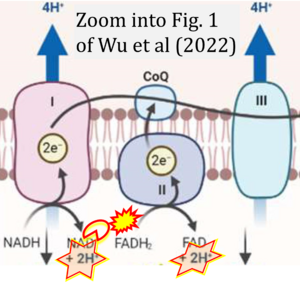
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Hydrogen ion ambiguities in the electron transfer system
Communicated by Gnaiger E (2023-10-08) last update 2023-11-10
- Electron (e-) transfer linked to hydrogen ion (hydron; H+) transfer is a fundamental concept in the field of bioenergetics, critical for understanding redox-coupled energy transformations.
- However, the current literature contains inconsistencies regarding H+ formation on the negative side of bioenergetic membranes, such as the matrix side of the mitochondrial inner membrane, when NADH is oxidized during oxidative phosphorylation (OXPHOS). Ambiguities arise when examining the oxidation of NADH by respiratory Complex I or succinate by Complex II.
- Oxidation of NADH or succinate involves a two-electron transfer of 2{H++e-} to FMN or FAD, respectively. Figures indicating a single electron e- transferred from NADH or succinate lack accuracy.
- The oxidized NAD+ is distinguished from NAD indicating nicotinamide adenine dinucleotide independent of oxidation state.
- NADH + H+ → NAD+ +2{H++e-} is the oxidation half-reaction in this H+-linked electron transfer represented as 2{H++e-} (Gnaiger 2023). Putative H+ formation shown as NADH → NAD+ + H+ conflicts with chemiosmotic coupling stoichiometries between H+ translocation across the coupling membrane and electron transfer to oxygen. Ensuring clarity in this complex field is imperative to tackle the apparent ambiguity crisis and prevent confusion, particularly in light of the increasing number of interdisciplinary publications on bioenergetics concerning diagnostic and clinical applications of OXPHOS analysis.