Lautrup 2019 Cell Metab
| Lautrup S, Sinclair DA, Mattson MP, Fang EF (2019) NAD+ in brain aging and neurodegenerative disorders. Cell Metab 30:630-55. https://doi.org/10.1016/j.cmet.2019.09.001 |
Lautrup S, Sinclair DA, Mattson MP, Fang Evandro F (2019) Cell Metab
Abstract: NAD+ is a pivotal metabolite involved in cellular bioenergetics, genomic stability, mitochondrial homeostasis, adaptive stress responses, and cell survival. Multiple NAD+-dependent enzymes are involved in synaptic plasticity and neuronal stress resistance. Here, we review emerging findings that reveal key roles for NAD+ and related metabolites in the adaptation of neurons to a wide range of physiological stressors and in counteracting processes in neurodegenerative diseases, such as those occurring in Alzheimer's, Parkinson's, and Huntington diseases, and amyotrophic lateral sclerosis. Advances in understanding the molecular and cellular mechanisms of NAD+-based neuronal resilience will lead to novel approaches for facilitating healthy brain aging and for the treatment of a range of neurological disorders.
• Bioblast editor: Gnaiger E
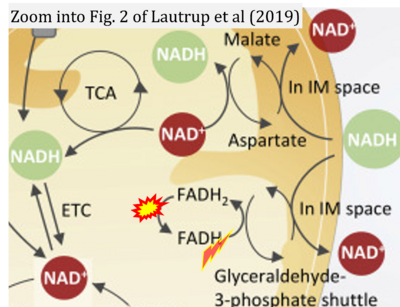
Correction: FADH2 and Complex II
- FADH2 is shown as the substrate feeding electrons into Complex II (CII). This is wrong and requires correction - for details see Gnaiger (2024).
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
- Fig. 2 of Lautrup et al (2019): FADH2/FAD does not reach the mt-matrix in the reaction catalyzed by mitochondrial glycerophosphate dehydrogenase. Nevertheless, FADH should be corrected to FAD.
- Gnaiger E (2024) Complex II ambiguities ― FADH2 in the electron transfer system. J Biol Chem 300:105470. https://doi.org/10.1016/j.jbc.2023.105470 - »Bioblast link«
Labels: Pathology: Aging;senescence, Neurodegenerative
NAD