Gonçalves 2019 Mitofit Preprint Arch EA
| Gonçalves Debora F, Courtes AA, Hartmann DD, Carvalho PR, Marques DM, Machado ML, Furtado AV, Soares FA, Dalla Corte CL (2019) Aging effects on mitochondrial control factors in Pink1 knockout Drosophila melanogaster. https://doi.org/10.26124/mitofit:ea19.MiPSchool.0002 |
» MitoFit Preprint Arch EA19.2.
Aging effects on mitochondrial control factors in Pink1 knockout Drosophila melanogaster
Goncalves DF, Courtes AA, Hartmann DD, Carvalho PR, Marques DM, Machado ML, Furtado AV, Soares FA, Dalla Corte CL (2019) MitoFit Preprint Arch
Abstract: Version 1 (v1) 2019-06-03 doi:10.26124/mitofit:ea19.MiPSchool.0002
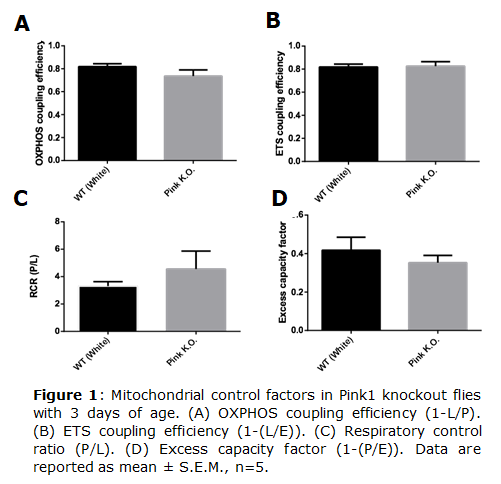
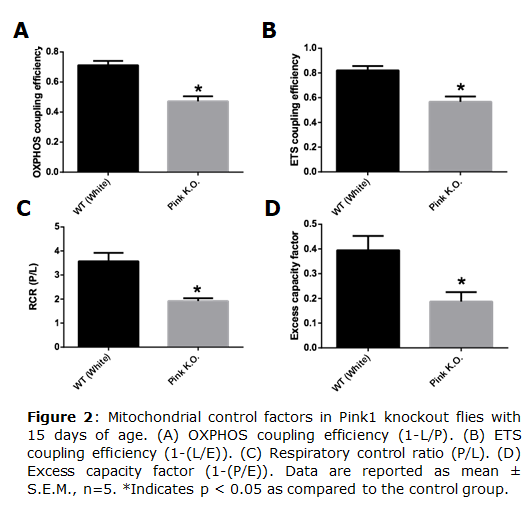
Parkinson disease (PD) is considered the second most common neurodegenerative disorder in the world and is related mainly with aging. PD is characterized by loss of dopaminergic neurons and formation of protein aggregates, such as α-synuclein [1], leading to motor dysfunction, and impairment of cognitive and memory functions [2]. The pathology and symptoms of PD are well described, although its mechanisms and causes remain unclear. One mechanism involved in PD is mitochondrial dysfunction [3]. Mutation in genes involved in mitochondrial quality control, such as PARK2 and Pink1, produce PD symptoms [4]. These genes code for proteins such as PINK1 (PTEN induced kinase 1) that is a serine/threonine kinase involved in mitochondrial network quality control [5]. In this work we evaluated mitochondrial quality control using Pink1 knockout of Drosophila melanogaster in different lifetime. • Keywords: PARK2 & PINK1 • Bioblast editor: Iglesias-Gonzalez J
Affiliations
Gonçalves Debora F(1), Courtes AA(1), Hartmann DD(1), Carvalho PR(1), Marques DM(1), Machado ML(1), Furtado AV(1), Soares FA(1), Dalla Corte CL(1,2) - [email protected]
- Univ Federal de Santa Maria, Brazil. Biochemistry and molecular biology Dpt.
- Univ Federaldo Pampa, Brazil
Results
References
- R. Requejo-Aguilar, I. Lopez-Fabuel, E. Fernandez, L.M. Martins, A. Almeida, J.P. Bolaños, PINK1 deficiency sustains cell proliferation by reprogramming glucose metabolism through HIF1., Nat. Commun. 5 (2014) 4514. doi:10.1038/ncomms5514.
- C.G. Goetz, The history of Parkinson’s disease: early clinical descriptions and neurological therapies., Cold Spring Harb. Perspect. Med. 1 (2011) a008862. doi:10.1101/cshperspect.a008862.
- N. Ammal Kaidery, B. Thomas, Current perspective of mitochondrial biology in Parkinson’s disease, Neurochem. Int. (2018). doi:10.1016/j.neuint.2018.03.001.
- H.-L. Wang, A.-H. Chou, A.-S. Wu, S.-Y. Chen, Y.-H. Weng, Y.-C. Kao, T.-H. Yeh, P.-J. Chu, C.-S. Lu, PARK6 PINK1 mutants are defective in maintaining mitochondrial membrane potential and inhibiting ROS formation of substantia nigra dopaminergic neurons., Biochim. Biophys. Acta. 1812 (2011) 674–84. doi:10.1016/j.bbadis.2011.03.007.
- C.A. Gautier, T. Kitada, J. Shen, Loss of PINK1 causes mitochondrial functional defects and increased sensitivity to oxidative stress, Proc. Natl. Acad. Sci. 105 (2008) 11364–11369. doi:10.1073/pnas.0802076105.
- D. Pesta, E. Gnaiger, High-Resolution Respirometry: OXPHOS Protocols for Human Cells and Permeabilized Fibers from Small Biopsies of Human Muscle, in: 2012: pp. 25–58. doi:10.1007/978-1-61779-382-0_3.
- R. Requejo-Aguilar, J.P. Bolaños, Mitochondrial control of cell bioenergetics in Parkinson’s disease, Free Radic. Biol. Med. 100 (2016) 123–137. doi:10.1016/j.freeradbiomed.2016.04.012.
Event
Preprints for Gentle Science
» MitoFit Preprints - the Open Access preprint server for mitochondrial physiology and bioenergetics
Labels:
Preprints